CAR-T Cell Therapy Market to Achieve USD 85.8 Billion by 2033, Driven by 32.0% CAGR | Dimension Market Research
Advancements and Projections in the CAR-T Cell Therapy Market: Insights, Trends, Opportunities, and Recent Developments
New York, June 10, 2024 (GLOBE NEWSWIRE) — Market Overview
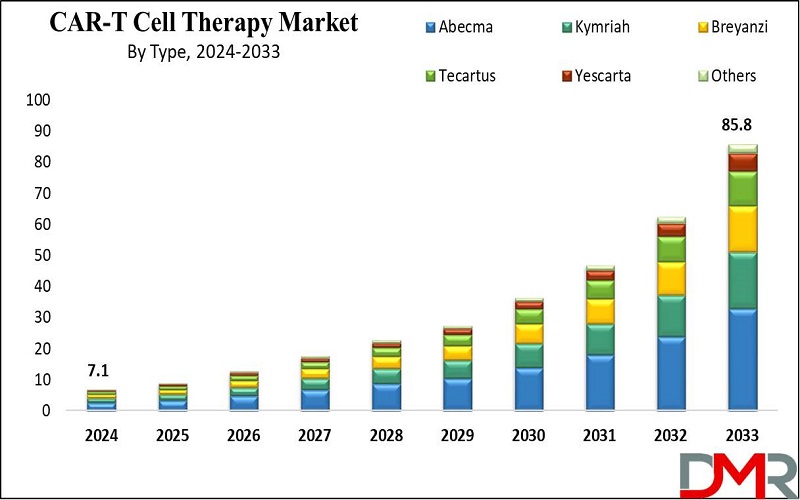
The Global CAR-T Cell Therapy Market size is expected to reach USD 7.1 billion by 2024 and is further anticipated to reach USD 85.8 billion by 2033 at a CAGR of 32.0% from 2024 to 2033.
CAR-T cell therapy modifies patient T-cells to target cancer, showing promise for blood cancers and under investigation for others. It involves lab-growing modified cells for infusion, driven by rising global cancer rates.
Click to Request Sample Report and Drive Impactful Decisions: https://dimensionmarketresearch.com/report/car-t-cell-therapy-market/request-sample/
The U.S. sees high new cancer cases and deaths, supporting CAR-T adoption. Regulatory approvals boost growth, despite side effects like Cytokine Release Syndrome and infection risks, posing safety concerns and necessitating effective management.
Important Insights
- Market Value: The global CAR-T cell therapy market is expected to grow significantly, reaching USD 85.8 billion by 2033, up from USD 7.1 billion in 2024.
- Market Definition: The CAR-T cell therapy market involves using living cells for treatment, focusing on regenerative medicine, immunotherapy, and personalized treatments, leading to improved medical outcomes and patient care.
- Type Segment Insights: Abecma is projected to dominate the CAR-T cell therapy market in 2024 with the highest market share and is expected to continue growing throughout the forecast period.
- Application Segment Insights: Leukemia is anticipated to lead the application segment, holding the largest market share in 2024 and maintaining its dominance in the CAR-T cell therapy market.
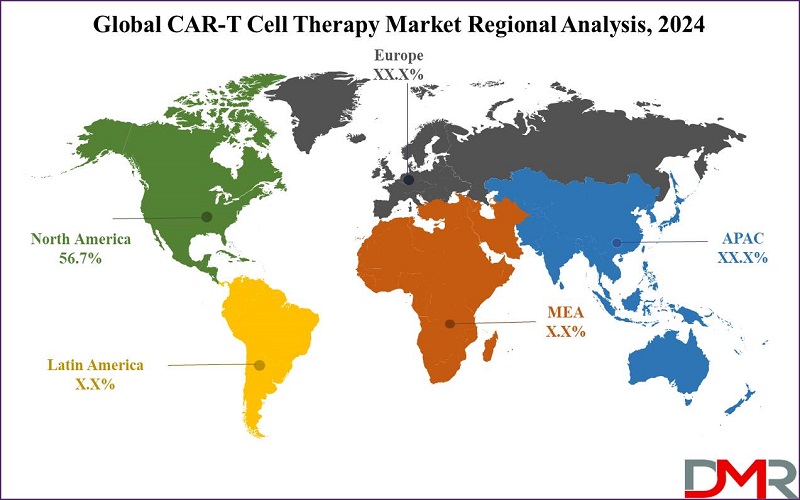
- Regional Analysis: North America is projected to account for 56.7% of the market revenue in 2024, continuing to dominate the global cell therapy market in the coming years.
Latest Trends
- Personalized Medicine in relation with the modern progress CAR-T cell therapy is also progressing in the sphere of personalized medicine and patient centric approaches to minimize the side effects and raise the effectiveness. New CAR-T cell products remain among the central points of competitive markets.
- This trend of companies and institutions’ collaboration facilitated by the expertise of biopharmaceutical firms and research organizations helps to fast-track the introduction of CAR-T cell therapies to the market.
CAR-T Cell Therapy Market: Competitive Landscape
- Global competition of the CAR-T cell therapy market is moderate and the large numbers of global manufacture execute such as partnership, product launch, expanding regional presence and mergers/acquisitions to enhance the position within the market. These dynamic approaches thus support development and industry influence.
- For instance, in June 2022 Bristol Myers Squibb claimed that FDA approved their Breyanzi as a safe and effective CAR-T cell therapy for adult patients with LBCL, meaning that this company has the necessary permits to treat this type of cancer.
Some of the prominent market players:
- Johnson & Johnson
- Novartis AG
- Eli Lilly & Company
- Celyad Oncology
- Bristol-Myers Squibb Company
- ACRO Biosystems
- Servier Laboratories
- Miltenyi Biotec
- Gilead Sciences Inc
- Sorrento Therapeutics Inc
- Other Key Players
Transform your business approach with strategic insights from our report. Get in touch to request our brochure today! : https://dimensionmarketresearch.com/report/car-t-cell-therapy-market/download-reports-excerpt/
CAR-T Cell Therapy Market Scope
| Report Highlights | Details |
| Market Size (2024) | USD 7.1 Bn |
| Forecast Value (2033) | USD 85.8 Bn |
| CAGR (2024-2033) | 32.0% |
| Leading Region in terms of Revenue Share | North America |
| Percentage of Revenue Share by Leading Region | 56.7% |
| Historical Data | 2017 – 2022 |
| Forecast Data | 2025 – 2033 |
| Base Year | 2023 |
| Estimate Year | 2024 |
| Segments Covered | By Type, By Target Antigen, By Application, By End User |
| Regional Coverage | North America, Europe, Asia Pacific, Latin America, Middle East & Africa (MEA) |
Market Analysis
Abecma is a CAR-T cell therapy that is recommended for the treatment of multiple myeloma by modifying patient T-cells to recognize proteins that are specific to the cancer. Though it has side effects it bare a hope for patients who have no many option due to its effectiveness in clinical trials. Abecma proves that CAR-T therapies still hold a vast potential for revolutionizing cancer treatment as other studies are being conducted for its augmentations.
Growth Drivers
- Growing Incidence of Cancer: The incidence of cancer, especially leukemia and lymphoma, is growing significantly and puts pressure on the market’s need for effective treatment options like CAR-T cell therapies.
- Increased Development in Gene Editing Technologies: Technological developments in the gene-editing techniques such as the CRISPR make CAR-T cell therapies safer and more efficient and reduce the total cost of production, thus, augmenting the overall market growth.
Restraints
- High Cost of Therapy: CAR-T cell therapy remains costly and therefore difficult to deploy in all countries, especially the developing ones that cannot afford the expensive treatments.
- Potential for Severe Side Effects: Possibility of High-Risk Side Effects, like cytokine release syndrome and neurotoxicity, need strict measures to control and could dissuade patients and healthcare providers hence affecting the market growth.
Growth Opportunities
- Expansion in North America: A high prevalence in cancer cases coupled with the growth of healthcare facilities in North America has been viewed as a sign of growth by the government through grants for cancer research.
- Emerging Markets in Asia-Pacific: The growth of the intensity of healthcare services and the increased focus on the fight against cancer offer significant opportunities for the development of the market for CAR-T cell therapy.
Purchase the Competition Analysis Dashboard Today: https://dimensionmarketresearch.com/checkout/car-t-cell-therapy-market/
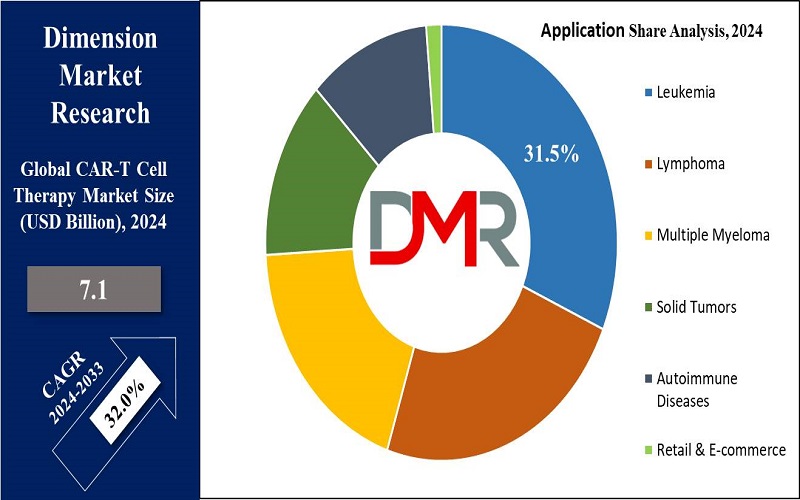
CAR-T Cell Therapy Market Segmentation
By Type
- Abecma Abecma (idecabtagene vicleucel)
- Kymriah (tisagenlecleucel)
- Breyanzi (lisocabtagene maraleucel)
- Tecartus (brexucabtagene autoleucel)
- Yescarta (axicabtagene ciloleucel)
- Others
By Target Antigen
- CD19
- BCMA (B-cell maturation antigen)
- CD20
- CD22
- Dual Targets (CD19/CD22, CD19/BCMA, etc.)
- Others
By Application
- Leukemia
- Acute Lymphocytic Leukemia (ALL)
- Chronic Lymphocytic Leukemia (CLL)
- Acute Myeloid Leukemia (AML)
- Lymphoma
- Diffuse Large B-cell lymphoma (DLBCL)
- Mantle Cell Lymphoma (MCL)
- Follicular Lymphoma (FL)
- Primary Mediastinal Large B-cell lymphoma (PMBCL)
- High-grade B-cell lymphoma (HGBL)
- Others
- Multiple Myeloma (MM)
- Solid Tumors
- Glioblastoma
- Neuroblastoma
- Others
- Autoimmune Diseases
- Others
By End User
- Hospitals
- Academic and Research Hospitals
- General Hospitals
- Specialized Cancer Hospitals
- Cancer Care Treatment Centers
- Clinical Research Organizations (CROs)
- Biotechnology and Pharmaceutical Companies
- Others
Click to Request Sample Report and Drive Impactful Decisions: https://dimensionmarketresearch.com/report/car-t-cell-therapy-market/request-sample/
Regional Analysis
North America is expected to leads the CAR-T cell therapy market with 56.7% revenue share in 2024, driven by strong research infrastructure, commercial activities, and numerous clinical studies. Increase in regulatory approval for drug and devices, and changes in reimbursement policy in US and Canada also fuel this growth.
The Asia Pacific and specifically China see their growth rates boosted by governmental investments and healthcare reforms, positioning this region as one of the main clinical trials and markets for CAR-T.
By Region
North America
- The U.S.
- Canada
Europe
- Germany
- The U.K.
- France
- Italy
- Russia
- Spain
- Benelux
- Nordic
- Rest of Europe
Asia-Pacific
- China
- Japan
- South Korea
- India
- ANZ
- ASEAN
- Rest of Asia-Pacific
Latin America
- Brazil
- Mexico
- Argentina
- Colombia
- Rest of Latin America
Middle East & Africa
- Saudi Arabia
- UAE
- South Africa
- Israel
- Egypt
- Rest of MEA
Discover additional reports tailored to your industry needs
- Handheld Surgical Instrument Market Reseach Report 2023
- Hairy Cell Leukemia Therapeutics Market Reseach Report 2023
- Genomics Personalized Health Market Reseach Report 2023
- Gene Synthesis Service Market Reseach Report 2023
- Gelatin-Based Plasma Expander Market Reseach Report 2023
- Gastroesophageal Reflux Disease (GERD) Therapeutics Market Reseach Report 2023
- Elapegademase-lvlr Market Reseach Report 2023
- EHR-EMR Market Reseach Report 2023
- E-Health Services Market Reseach Report 2023
- Dietary Supplement Market Reseach Report 2023
Recent Developments in the CAR-T Cell Therapy Market
- November 2023: Selecta Biosciences merged with Cartesian Therapeutics, securing USD 110 million to develop Descartes-08, an RNA-engineered CAR-T therapy for MG, and other RNA cell therapy programs.
- December 2023: Bristol-Myers Squibb received approval for Abecma’s new indication, a BCMA-directed CAR-T therapy for relapsed or refractory multiple myeloma patients who have undergone prior treatments.
- September 2023: 2seventy Bio and JW Therapeutics announced plans to expand their alliance, adding two new candidates for solid tumors using TCR technology and for autoimmune diseases with CAR-T approaches.
- May 2023: Autolus Therapeutics’ Phase 2 FELIX study of obe-cel for relapsed/refractory adult B-cell Acute Lymphoblastic Leukemia was selected for an oral presentation at the EHA 2023 Congress.
- June 2022: Bristol Myers Squibb received FDA approval for Breyanzi, a CD19-directed CAR-T therapy for treating adult patients with large B-cell lymphoma.
- April 2022: Autolus Therapeutics collaborated with Cardinal Health to support the U.S. launch and commercialization of its CAR-T therapy, pending FDA approval.
- April 2022: Kite, a Gilead Company, received FDA approval for Yescarta CAR-T therapy for adult patients with large B-cell lymphoma that is refractory to or relapses after first-line chemoimmunotherapy.
About Dimension Market Research (DMR):
Dimension Market Research (DMR) is a market research and consulting firm based in India & US, with its headquarters located in the USA (New York). The company believes in providing the best and most valuable data to its customers using the best resources analysts work, to create unmatchable insights into the industries, and markets while offering in-depth results of over 30 industries, and all major regions across the world.
We also believe that our clients don’t always want what they see, so we provide customized reports as well, as per their specific requirements to create the best possible outcomes for them and enhance their business through our data and insights in every possible way.
CONTACT: Global Business Development Team United States 957 Route 33, Suite 12 #308 Hamilton Square, NJ-08690 Phone No.: +1 732 369 9777, +91 88267 74855 Inquiry@dimensionmarketresearch.com
 Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. SingaporeOutlook.com takes no editorial responsibility for the same.
Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. SingaporeOutlook.com takes no editorial responsibility for the same.

