Global Point-of-Care Molecular Diagnostics Market Projected to Reach USD 13.05 Billion by 2035; Growth is Driven by Increasing Need for Fast Near-Patient Testing Globally – SNS Insider
Growing Adoption of PCR-Based Platforms, Isothermal Amplification Technologies, and Lab-on-a-Chip Systems Accelerating Market Expansion Globally.
Austin, United States, March 09, 2026 (GLOBE NEWSWIRE) — Point-of-Care Molecular Diagnostics Market Size & Growth Analysis:
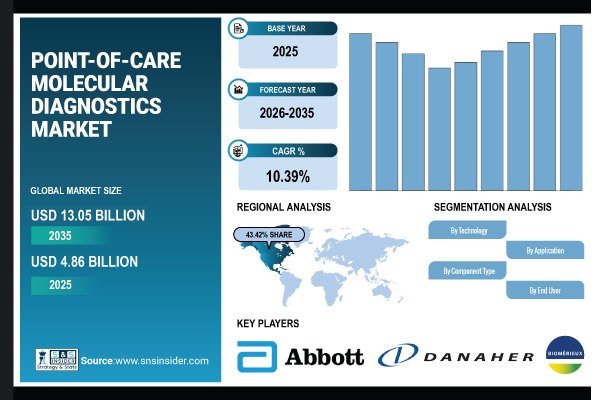
According to SNS Insider, The Point-of-Care Molecular Diagnostics Market size was valued at USD 4.86 Billion in 2025 and is expected to reach USD 13.05 Billion by 2035, growing at a CAGR 10.39% during 2026-2035. The rise in need for fast near-patient testing and early disease diagnosis in hospitals and clinics, as well as within the home, is considerably speeding up the Point-of-Care Molecular Diagnostics market.
Market Size and Forecast
- Market Size in 2025: USD 4.86 Billion
- Market Size by 2035: USD 13.05 Billion
- CAGR: 10.39%
- Base Year: 2025
- Forecast Period: 2026-2035
- Historical Data: 2022-2024
Get a Sample Report of Point-of-Care Molecular Diagnostics Market: https://www.snsinsider.com/sample-request/9763
At a compound annual growth rate (CAGR) of 5.63%, the U.S. point-of-care molecular diagnostics market is expected to increase from USD 1.72 billion in 2025 to USD 3.48 billion by 2035. The expansion of home and outpatient testing, growing demand for quick near-patient testing, rising rates of infectious and chronic diseases, ongoing developments in PCR, isothermal amplification, and lab-on-a-chip technologies, and growing R&D and commercialization of POC molecular diagnostic solutions are all factors driving growth.
Rising Demand for Rapid Near-Patient Testing and Early Disease Detection to Augment Market Growth Globally
Increased military spending, defense modernization initiatives, and the strategic need for force multipliers all contribute to market expansion. Loyal-wingman drones, AI systems, and other autonomous combat technologies simplify operations, lower pilot risk, and enable the completion of tasks that are too risky for humans. Adoption by air forces worldwide is accelerating due to rising R&D expenditures, the development of advanced sensor fusion, and the emphasis on network-centric combat.
High Development Costs, Technical Complexity, and Regulatory Challenges May Limit Market Expansion Globally
The growing use of POC molecular diagnostics in home care settings, ambulatory facilities, and emerging and resource-constrained areas will be the primary driver of market expansion. Market penetration should continue to rise because to the rising technological sophistication of multiplex testing, smaller equipment, and integration with digital health systems. Through 2035, advancements in more affordable, portable, and highly accurate point-of-care molecular systems are expected to boost global revenue growth and usage.
Major Players Analysis Listed in this Report are:
- Abbott Laboratories
- Roche Diagnostics
- Danaher Corporation (Cepheid)
- bioMérieux SA
- QIAGEN N.V.
- Thermo Fisher Scientific
- QuidelOrtho Corporation
- Hologic
- Becton Dickinson (BD)
- Bio-Rad Laboratories
- Meridian Bioscience
- Biocartis Group NV
- Co-Diagnostics
- Binx Health
- Molbio Diagnostics Pvt. Ltd.
- GenMark Diagnostics
- Siemens Healthineers
- Orasure Technologies
- Visby Medical
- AccuBioTech Co. Ltd.
Segmentation Analysis:
By Technology
In 2025, PCR-based tests dominated with 50% share due to high accuracy, reproducibility and widespread adoption in hospitals, clinics, and laboratory environments. The largest growth in tests is being seen for isothermal amplification-based tests, due to the growing need for rapid, portable and inexpensive point-of-care molecular diagnostics.
By Application
In 2025, infectious diseases testing dominated with 45% share as high-speed examination of infectious pathogens such as COVID-19, influenza, HIV, and TB is crucial for timely clinical decisions and outbreak management. The Oncology testing segment will grow at the fastest CAGR during the forecast period, and high-volume demand for early cancer detection along with growing interest in genetic biomarkers screening and personalized medicine are boosting segment growth.
By End-Use
In 2025, hospitals held 55% share as hospitals are the main centers for clinical diagnostics providing adequately trained personnel, standard workflows, and infrastructure that assures regulatory compliance. Among the end-use segments, home testing is anticipated to be the fastest growing segment due to increasing preference for rapid and self-testing of molecular tests by patients.
By Component Type
In 2025, cartridge-based disposable kits led with 48% share as it is highly preferred due to ease of use, similar workflows, and consistent sample handling. The fastest growing segment is lab-on-a-chip platforms, the ability to mix multiple tests in one place, with rapid turnaround and portable nature.
Need Any Customization Research on Point-of-Care Molecular Diagnostics Market, Enquire Now: https://www.snsinsider.com/enquiry/9763
Point-of-Care Molecular Diagnostics Market Segmentation
By Technology
- PCR-based Tests
- Isothermal Amplification-based Tests
- Sequencing-based tests
By Application
- Infectious Diseases
- Oncology
- Antimicrobial Resistance Testing
By End Use
- Hospitals
- Home Testing
- Ambulatory Facilities
By Component Type
- Cartridge-based Disposable kits
- Portable Instruments
- Lab-on-a-chip Platforms
Regional Insights:
The shares of the North America market reached 43.42% in 2025, ranking the first in the global market. This is attributed to the presence of advanced healthcare infrastructure, high adoption of rapid diagnostic testing, and strong presence of leading POC molecular diagnostics companies in the region. The U.S. and Canada are top contenders for tech development, regulatory approvals, and early adoption of advanced POC systems.
Asia-Pacific occupies a rapidly growing site in the Point-of-Care Molecular Diagnostics market, and is expected to grow with a CAGR of 15.82% during 2026-2035. The region is projected to witness the fastest growth due to the increasing usage of home testing, outpatient clinics, and decentralized healthcare services.
Recent Developments:
- In 2025, Abbott announced its planned acquisition of Exact Sciences for up to USD 23 billion, marking a major expansion into cancer screening and broader molecular diagnostics capabilities, strengthening its diagnostic portfolio and long‑term growth potential.
- In 2025, Roche received FDA 510(k) clearance and CLIA waiver for its cobas Liat STI multiplex assay panels, enabling rapid decentralized testing for sexually transmitted infections and expanding its POC molecular diagnostics offerings.
Exclusive Sections of the Report (The USPs):
- ADOPTION & DEPLOYMENT METRICS – helps you understand the adoption of point-of-care molecular diagnostics across hospitals, clinics, and home testing settings along with deployment trends across infectious disease testing, oncology and genetic screening, and antimicrobial resistance monitoring.
- PRODUCT & FUNCTIONAL INTEGRATION – helps you analyze preference patterns for PCR-based tests, isothermal amplification technologies, and sequencing-based POC platforms along with their integration within portable analyzers, cartridge-based kits, and lab-on-a-chip diagnostic systems.
- QUALITY & REGULATORY COMPLIANCE METRICS – helps you evaluate product performance expectations across hospitals, ambulatory clinics, and home testing environments along with increasing compliance requirements related to regulatory approvals, ISO standards, and laboratory accreditation guidelines.
- TECHNOLOGICAL ADOPTION RATE – helps you uncover the adoption of miniaturized diagnostic devices, automated sample processing technologies, and multiplex testing systems enabling faster and more accurate point-of-care molecular diagnostics.
- SUPPLY CHAIN DISRUPTION INDEX – helps you identify supply chain reliability trends, component standardization requirements, and strategic partnerships between diagnostic developers and healthcare providers influencing global deployment.
- COMPETITIVE LANDSCAPE – helps you gauge the competitive strength of key players based on their technological capabilities, diagnostic platform portfolios, strategic collaborations, and recent innovations in rapid molecular testing solutions.
Point-of-Care Molecular Diagnostics Market Report Scope
| Report Attributes | Details |
| Market Size in 2025 | USD 4.86 Billion |
| Market Size by 2035 | USD 13.05 Billion |
| CAGR | CAGR of 10.39% From 2026 to 2035 |
| Base Year | 2025 |
| Forecast Period | 2026-2035 |
| Historical Data | 2022-2024 |
| Regional Analysis/Coverage | North America (US, Canada), Europe (Germany, UK, France, Italy, Spain, Russia, Poland, Rest of Europe), Asia Pacific (China, India, Japan, South Korea, Australia, ASEAN Countries, Rest of Asia Pacific), Middle East & Africa (UAE, Saudi Arabia, Qatar, South Africa, Rest of Middle East & Africa), Latin America (Brazil, Argentina, Mexico, Colombia, Rest of Latin America). |
Purchase Single User PDF of Point-of-Care Molecular Diagnostics Market Report (20% Discount): https://www.snsinsider.com/checkout/9763
Table of Contents – Major Key Points
1. Introduction
2. Executive Summary
3. Market Overview
4. Statistical Insights & Trends Reporting
5. Point-of-Care Molecular Diagnostics Market Segmental Analysis & Forecast, By Technology
6. Point-of-Care Molecular Diagnostics Market Segmental Analysis & Forecast, By Application
7. Point-of-Care Molecular Diagnostics Market Segmental Analysis & Forecast, By End Use
8. Point-of-Care Molecular Diagnostics Market Segmental Analysis & Forecast, By Component Type
9. Point-of-Care Molecular Diagnostics Market Segmental Analysis & Forecast, By Region
10. Competitive Landscape
11. Analyst Recommendations
12. Assumptions
13. Disclaimer
14. Appendix
Access Complete Report Details of Point-of-Care Molecular Diagnostics Market Analysis & Outlook: https://www.snsinsider.com/reports/point-of-care-molecular-diagnostics-market-9763
[For more information or need any customization research mail us at info@snsinsider.com]
About Us:
SNS Insider is one of the leading market research and consulting agencies that dominates the market research industry globally. Our company’s aim is to give clients the knowledge they require in order to function in changing circumstances. In order to give you current, accurate market data, consumer insights, and opinions so that you can make decisions with confidence, we employ a variety of techniques, including surveys, video talks, and focus groups around the world.
CONTACT: Contact Us: Rohan Jadhav - Principal Consultant Phone: +1-315 636 4242 (US) | +44- 20 3290 5010 (UK) Email: info@snsinsider.com
 Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. SingaporeOutlook.com takes no editorial responsibility for the same.
Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. SingaporeOutlook.com takes no editorial responsibility for the same.

