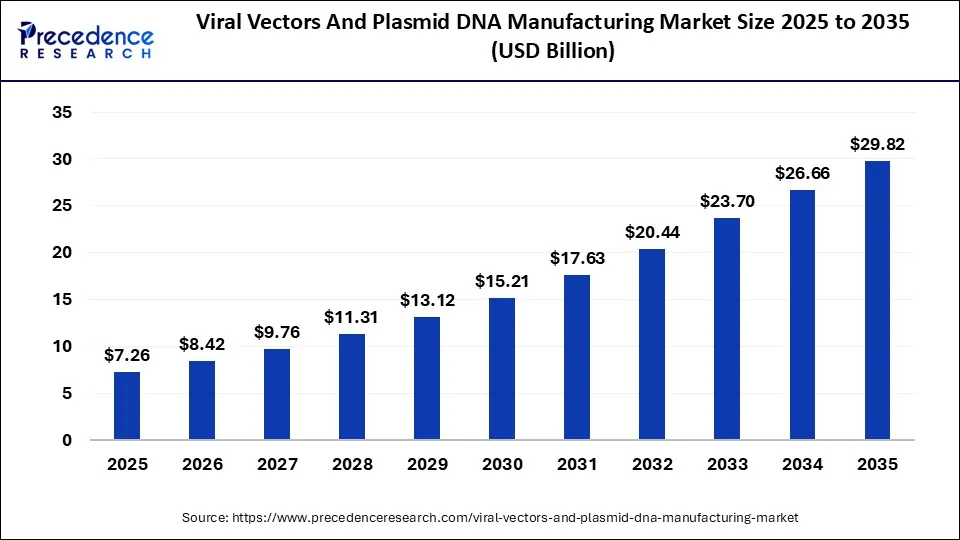
Viral Vectors & Plasmid DNA Manufacturing Market Size Worth USD 29.82 Bn by 2035 Growing Demand for Gene and Cell Therapies Drives the Market
The globe viral vectors & plasmid DNA manufacturing market size is expected to be lead USD 29.82 billion by 2035, increasing from USD 7.26 billion in 2025, with a strong CAGR of 15.17% from 2026 to 2035.
Ottawa, April 20, 2026 (GLOBE NEWSWIRE) — According to Precedence Research, the global viral vectors & plasmid DNA manufacturing market size will grow from USD 8.42 billion in 2026 to nearly USD 29.82 billion by 2035, accelerating with a solid CAGR of 15.17% from 2026 to 2035. The viral vectors and plasmid DNA manufacturing market is expanding rapidly due to rising cell and gene therapy development, increasing clinical trials, and demand for scalable biomanufacturing, supported by technological advancements and growing biotech investments worldwide.
The Complete Study is Now Available for Immediate Access | Download the Sample Pages of this Report@ https://www.precedenceresearch.com/sample/1012
Viral Vectors and Plasmid DNA Manufacturing Market Key Takeaways
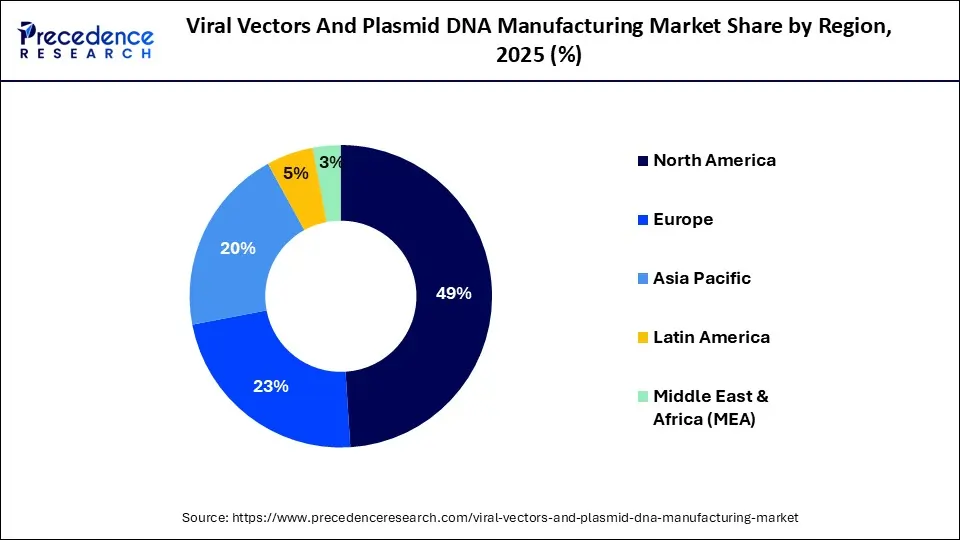
- In 2025, North America led the viral vectors and plasmid DNA manufacturing market, contributing 49% of the total revenue.
- Among vector types, the AAV segment emerged as a key growth driver, accounting for a 21% revenue share in 2025.
- Based on workflow, downstream processing held a dominant position, capturing 54% of the market revenue in 2025.
- By application, vaccinology represented the largest share, contributing approximately 22.5% of the total revenue in 2025.
- In terms of disease focus, the cancer segment dominated with a 38% revenue share in 2025.
- By end use, research institutes led the market, securing around 58.4% of the total revenue in 2025.
➡️ Become a valued research partner with us ☎ https://www.precedenceresearch.com/schedule-meeting
Why Do There is an Increase in Demand for Viral Vectors and Plasmid DNA Manufacturing?
Both the viral vectors and plasmid DNA manufacturing sector of the market has seen accelerated growth as there has been more gene and cell therapy products in development that target cancer and rare diseases. As approvals are granted and there continues to be more late-stage clinical trials this has increased the demand for high-quality products in the vector production market. New advancements in gene-editing technologies such as CRISPR and mRNA-based platforms will continue to fuel the demand for scalable manufacturing of plasmid DNA.
Additionally, we have seen an increase in collaborations between biopharmaceutical companies and contract development and manufacturing organizations that have increased their production capacity and decreased their turn-around times. Innovative bioprocessing technologies and automation are significantly increasing production efficiency and investment from both the government and private sectors continues to support innovation and expanding infrastructure globally.
Market Opportunity
The viral vectors-plasmid DNA (pDNA) market is poised for strong future opportunities largely driven by the growth of both the gene/cell therapy development pipeline and the entrance of gene/cell therapies into the marketplace via regulatory approval. Between now and 2025, 8–10 new gene therapies are anticipated to be approved annually based on the FDA’s recent expedited approvals, while the currently-expanding number of late-stage clinical trials will also drive demand for dependable large-scale manufacturing. Innovations like the utilization of single-use bioreactors and advancements in purification systems afford manufacturers the opportunity for more flexible manufacturing with increased efficiency.
In addition, advancements in artificial intelligence (AI) based vector design; the construction of new cGMP-compliant manufacturing facilities; and other recent improvements in the development of vectors will continue to improve development and commercialization times for all areas of gene therapy development. As a result, applications for both oncology, rare genetic disease, and next-generation vaccines will afford additional opportunity for novel vector manufacturers and contract development organizations (CDO).
Built for leaders who move markets. Access live, actionable intelligence with Precedence Q. https://www.precedenceresearch.com/precedenceq/
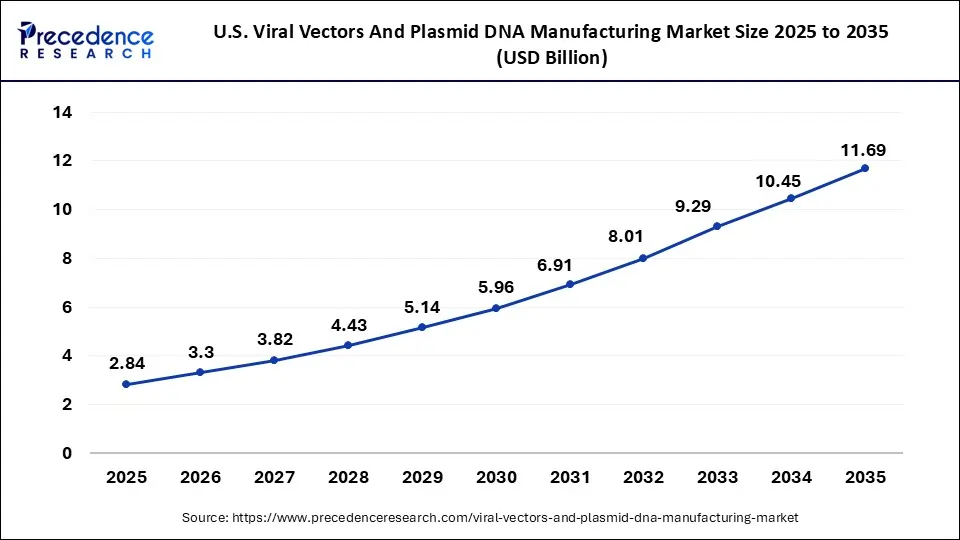
How Big is the Size of U.S. Viral Vectors & Plasmid DNA Manufacturing Market in 2026?
According to Precedence Research, the U.S. viral vectors and plasmid DNA manufacturing market size is valued at USD 3.3 billion in 2026 and is predicted to reach around USD 11.69 billion by 2035, growing at a significant CAGR of 15.2% from 2026 to 2035.
Note: This report is readily available for immediate delivery. We can review it with you in a meeting to ensure data reliability and quality for decision-making.
Try Before You Buy – Get the Sample Report@ https://www.precedenceresearch.com/sample/1012
Why Does North America Dominate The Market?
The viral vectors and the plasmid DNA manufacturing market is dominated by North America because of the regions significant share of established and expanding biopharmaceutical companies, including companies such as Thermo Fisher Scientific and Catalent. North America also has substantial investments being made into gene therapies, mRNA technology and cell-based research. The growing number of clinical trials, as well as strong government support of advanced therapies has increased the demand for high-quality vector manufacturing. Moreover, companies and contract manufacturers developing vectors are continuously adding new manufacturing facilities, partnering together to enhance their production capacity and scalability to commercialize innovative therapies more quickly.
Why Is Asia Pacific is the Fastest-Growing Region in the Vectors and Plasmid DNA Manufacturing Market?
The Middle East & Africa are enjoying the greatest growth in the petrochemicals industry as a result of increasing investments and abundance of available raw materials (i.e. feedstock). Individual nations are actively working to reduce their dependence on exporting crude by growing their downstream petrochemical production. New integrated facilities are coming on-stream, and strategic partnerships are forming between the nations of this region and major international companies. Growth will continue, as a result of increased consumption of plastics, fertilisers, & specialty chemicals, and from the development of infrastructure. Additionally, the MENA region’s location will continue to serve it well as a great platform from which to export to both Asia & Europe, & improve the competitiveness of MENA region in global markets.
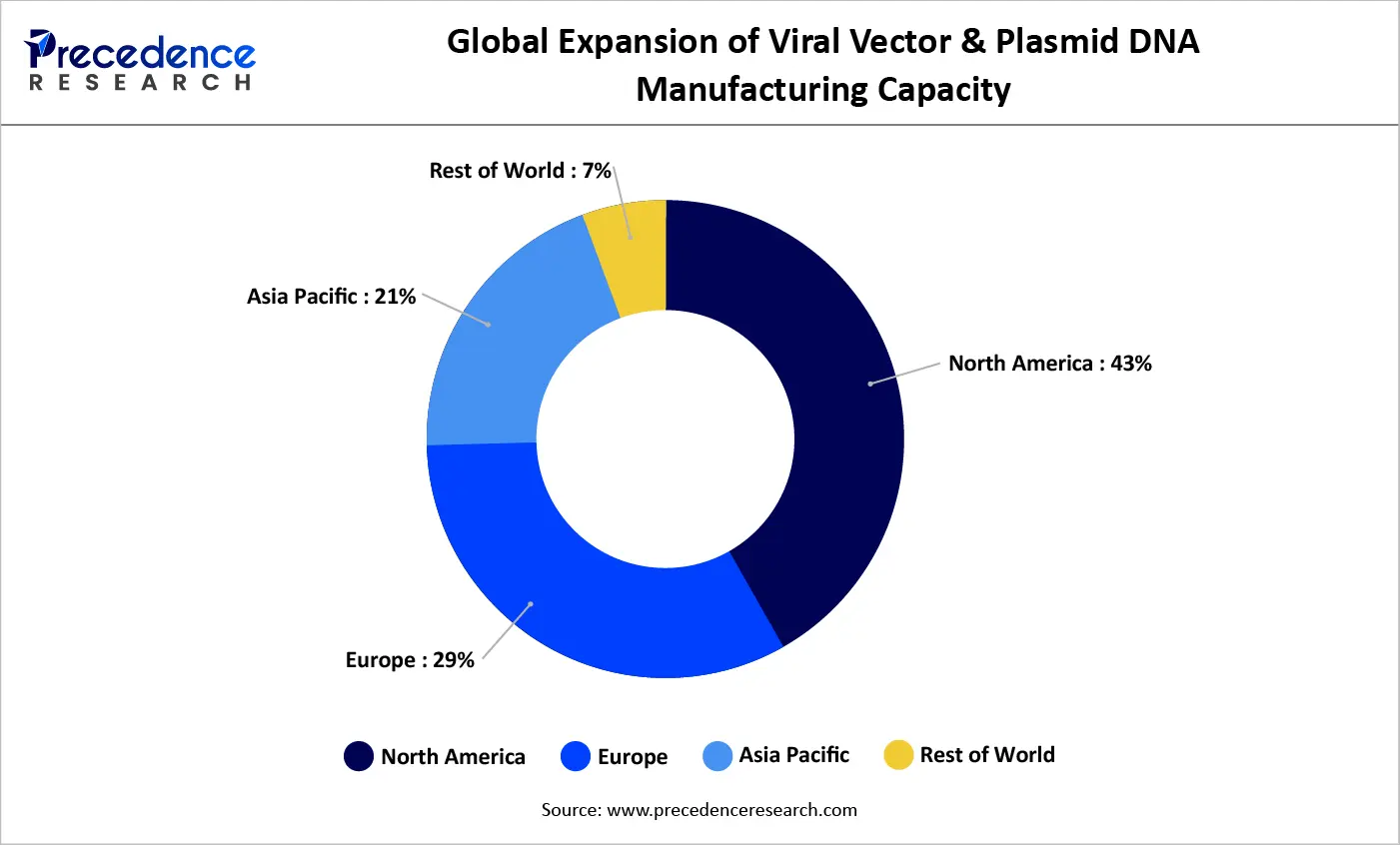
Global Manufacturing Capacity Expansion for Viral Vectors and Plasmid DNA (2021–2025)
From 2021 to 2025, global manufacturing capacity for viral vectors and plasmid DNA has grown consistently, driven by increasing demand from gene therapies, cell therapies, and mRNA-based treatments. North America remains the leading region, with its share rising from 40% to 43%, supported by strong biopharmaceutical investments and a well-established contract manufacturing network.
Europe has also expanded its footprint, with capacity increasing from 29% to 30%, fueled by government support and investments in GMP-compliant facilities. Meanwhile, Asia Pacific is witnessing the fastest growth, with its share climbing from 18% to 21%, driven by rising clinical trial activity and efforts to localize manufacturing.
In contrast, the rest of the world has experienced a relative decline, highlighting the continued concentration of advanced manufacturing capabilities within established biotechnology hubs.
Get informed with deep-dive intelligence on AI’s market impact https://www.precedenceresearch.com/ai-precedence
Viral Vectors & Plasmid DNA Manufacturing Market Insights
| Report Metrics | Details |
| Market Size in 2025 | USD 7.26 Billion |
| Market Size in 2026 | USD 8.42 Billion |
| Market Size by 2035 | USD 29.82 Billion |
| Market Growth (2026 – 2035) | 15.17% CAGR |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Vector Type, Application, Workflow, End-User, Disease, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
➤ Access the Full Viral Vectors & Plasmid DNA Manufacturing Market Study@ https://www.precedenceresearch.com/viral-vectors-and-plasmid-dna-manufacturing-market
Segmental Insights
Vector Type Insights
Why Are Adenovirus vectors Dominated the Market in 2025?
Adenovirus vectors leads the viral vectors & plasmid DNA manufacturing market. These vectors can effectively transduce both dividing and non-dividing cells, making them applicable to a wide range of areas such as gene delivery, vaccine development, and stimulating immune responses. Their established manufacturing processes and ability to scale up have made them leaders in the therapeutic and research arena.
The lentivirus vectors are growing rapidly, due to their ability to integrate into the host genome, which allows for prolonged expression of the gene. They are becoming increasingly used among advanced therapies, such as gene and cell therapy, as well as rising in popularity for use in precision medicine and growing numbers of clinical trials in biopharmaceuticals.
Workflow Insights
Which Workflow Is Leading the Viral Vectors & Plasmid DNA manufacturing Market?
The downstream processing segment dominated the market in 2025. The majority of the workflow processes relevant to viral vector production occurs during downstream processing; therefore, they are critical for achieving product purity, safety, and quality. This includes the most critical steps in the validation process such as purification and fill/finish, as well as many product stability issues. The growing emphasis on the production of high-quality biopharmaceuticals and the increased need for compliance with strict manufacturing regulations have increased the importance of downstream processing to the large-scale manufacture of biopharmaceuticals worldwide.
The upstream processing is experiencing rapid growth as a direct consequence of increased demand for efficient production and scalable production of vectors. Advances have also been made in cell culture technology and significant developments in bioreactor systems have driven the increase of overall productivity in upstream processes. Demand for quantity and cost-effectiveness within the production of vectors continues to encourage innovative approaches to increase the generated quantity of vector.
End User Insights
Why did the Research Institute Segment Dominate the Market?
Research institutes comprise the largest market segment within the viral vectors & plasmid DNA manufacturing market based on end-user. Research institutes support extensive research and development (R&D) for new therapies that utilize viral vectors and consequently produce the largest quantity of new research and preclinical and clinical data within the viral vector space. The continuous R&D performed by research institutes is supported by both government funding and non-profit partnerships.
The biopharmaceutical and pharmaceutical sectors are the fastest-growing segment, driven by both the increasing amounts of capital being invested into the commercialization of gene and cell therapy products, as well as the rapid expansion of their manufacturing capabilities and portfolio of products. The rise of advanced therapeutics within these sectors has led to increased use of viral vectors in these sectors.
By Disease Insights
Cancer dominates this segment due to its high incidence and increasing demand for targeted treatments. Gene transfer and Immunotherapies utilize viral vectors for the delivery of gene therapeutics to help treat cancer patients and continuous active research & clinical development provides greater support for the utilization of Viral Vectors to ultimately provide effective treatments for cancer.
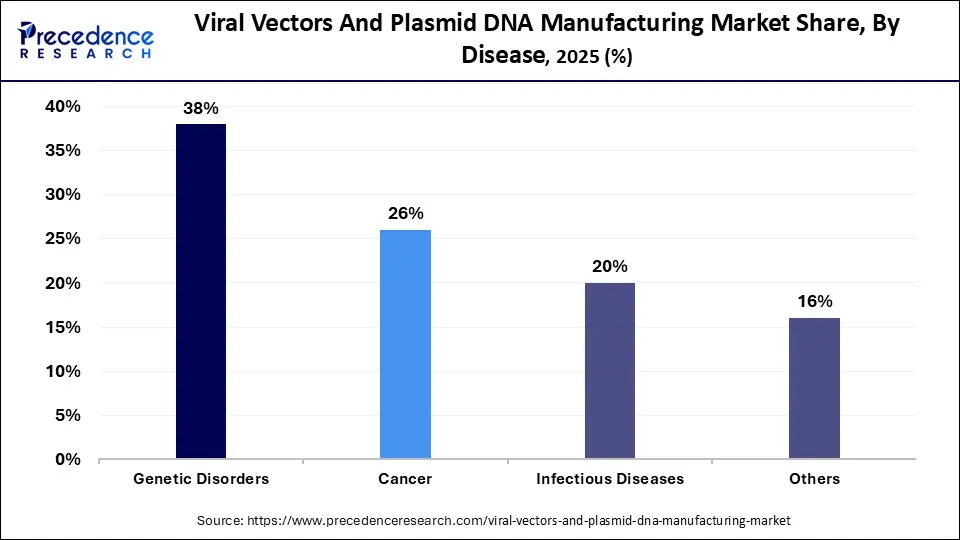
The fastest growing segment is Genetic Disorders. The continuing increased emphasis on rare disease therapies and gene therapy has resulted in the growth of this segment. The use of viral vectors is highly appropriate for precision genetic modifications, which is the hallmark of rare genetic diseases, and growing awareness, better diagnostics and further advancement in gene therapies are driving even faster growth in this segment.
✚ Related Topics You May Find Useful:
➡️ Viral Vectors-Based Gene Therapy for Non-Human Primates Market: Explore advancements in preclinical models accelerating gene therapy innovation
➡️ Viral Vectors for Non-Human Primates Market: Understand growing demand for translational research in neuroscience and genetic studies
➡️ U.S. Plasmid DNA Manufacturing Market: Analyze expansion driven by gene therapy and mRNA production capabilities
➡️ U.S. Targeted DNA RNA Sequencing Market: Discover precision diagnostics trends shaping next-generation sequencing adoption
➡️ DNA Synthesis Market: Track innovations enabling synthetic biology and advanced genetic engineering
➡️ DNA Polymerase Market: Examine rising applications in PCR, sequencing, and molecular diagnostics
➡️ DNA Ligases Market: Explore key enzymes driving breakthroughs in genetic research and biotechnology
➡️ DNA Diagnostics Market: Understand the growing role of genetic testing in personalized medicine
➡️ DNA Synthesizer Market: Analyze demand for automated platforms supporting rapid DNA production
➡️ DNA Data Storage Market: Discover emerging solutions for ultra-high-density and long-term data storage
What are the Major Developments in the Viral Vectors and Plasmid DNA Manufacturing Market?
- In July 2025, ProBio opened a 128,000 sq. ft. flagship GMP facility in Hopewell, New Jersey, dedicated to plasmid DNA and viral vector manufacturing, strengthening cell and gene therapy production capabilities and accelerating clinical-stage development.
- In May 2025, 3P Biovian introduced two advanced platforms for AAV and plasmid DNA manufacturing, designed to streamline scalable production, improve process efficiency, and address increasing global demand for high-quality gene therapy materials.
Competitive Landscape

- Novasep
- Aldevron
- Creative Biogene
- The Cell and Gene Therapy Catapult
- Cobra Biologics
Segments Covered in the Report
By Vector Type
- Adenovirus
- Plasmid DNA
- Lentivirus
- Retrovirus
- AAV
- Others
By Application
- Gene Therapy
- Antisense &RNAi
- Cell Therapy
- Vaccinology
By Workflow
- Upstream Processing
- Vector Recovery/Harvesting
- Vector Amplification & Expansion
- Downstream Processing
- Fill-finish
- Purification
By End-User
- Biopharmaceutical and Pharmaceutical Companies
- Research Institutes
- By Disease
- Genetic Disorders
- Cancer
- Infectious Diseases
- Others
By Geography
- North America
- Asia Pacific
- Europe
- Latin America
- Middle East & Africa
Thank you for reading. You can also get individual chapter-wise sections or region-wise report versions, such as North America, Europe, or Asia Pacific.
Immediate Delivery Available | Buy This Premium Research Report@ https://www.precedenceresearch.com/checkout/1012
You can place an order or ask any questions, please feel free to contact at sales@precedenceresearch.com | +1 804 441 9344
Stay Ahead with Precedence Research Subscriptions
Unlock exclusive access to powerful market intelligence, real-time data, and forward-looking insights, tailored to your business. From trend tracking to competitive analysis, our subscription plans keep you informed, agile, and ahead of the curve.
Browse Our Subscription Plans@ https://www.precedenceresearch.com/get-a-subscription
About Us
Precedence Research is a worldwide market research and consulting organization. We give an unmatched nature of offering to our customers present all around the globe across industry verticals. Precedence Research has expertise in giving deep-dive market insight along with market intelligence to our customers spread crosswise over various undertakings. We are obliged to serve our different client base present over the enterprises of medicinal services, healthcare, innovation, next-gen technologies, semi-conductors, chemicals, automotive, and aerospace & defense, among different ventures present globally.
Web: https://www.precedenceresearch.com
Our Trusted Data Partners:
Towards Healthcare | Towards Packaging | Towards Chem and Materials | Towards FnB | Statifacts | Nova One Advisor | Market Stats Insight
Get Recent News:
https://www.precedenceresearch.com/news
For the Latest Update Follow Us:
LinkedIn | Medium | Facebook | Twitter
 Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. SingaporeOutlook.com takes no editorial responsibility for the same.
Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. SingaporeOutlook.com takes no editorial responsibility for the same.

